Putting Neglected Tropical Diseases in the Spotlight: Lessons Learned from Chagas Disease - State of the Planet
Putting Neglected Tropical Diseases in the Spotlight: Lessons Learned from Chagas Disease - State of the Planet |
| Posted: 30 Jan 2020 02:17 PM PST Putting Neglected Tropical Diseases in the Spotlight: Lessons Learned from Chagas Disease Photos of a "kissing bug," whose feces can spread the parasite that causes Chagas disease. Photo: CDC Today, January 30th 2020, is the inaugural World Neglected Tropical Diseases day. For the first time, global community leaders, health experts, civil society advocates, and policymakers have united to raise awareness about neglected tropical diseases (NTDs), in hopes of removing the "neglected" label from the group's name. But what are these NTDs, and why are they neglected? NTDs are a mixed group of parasitic and bacterial diseases that disproportionately affect impoverished and under-represented minority groups around the world. Some examples of these diseases include soil-transmitted helminthiasis (caused by parasitic worms such as hookworm), trachoma, Chagas disease, leishmaniasis, and schistosomiasis, among others. These diseases affect 1.5 billion people, so they are not rare diseases, just hidden in the local and global agendas. Moreover, although NTDs cause a high disease burden in low- and middle- income countries, they also impose an under-appreciated burden in wealthier nations. The World NTD day can help lift the veil, to not only advocate for political and financial commitments in the battle against NTDs, but also celebrate the successes achieved so far in reducing the burden of these diseases. The United Nations' 2030 Agenda for Sustainable Development includes promoting human health and well-being globally, recognizing the clear link between human health and sustainability. Poverty is considered the main structural determinant of NTDs because of its influence on living conditions and access to health services. Moreover, the burden created by these diseases feeds back into the social and economic situation, and combines with other determinants that generate health inequalities (such as gender and ethnicity) to keep the affected individuals and demographic groups in a poverty trap. Here, I want to bring the attention to Chagas disease, which was the focus of my dissertation research. Chagas disease is one of the main NTDs affecting vulnerable communities in Latin America. Mild cases include swelling, fever, diarrhea, and vomiting. Longer-lasting cases can cause heart failure and cardiac arrest. Chagas disease is caused by a parasite called Trypanosoma cruzi and it is transmitted by insect vectors from the subfamily Triatominae. Through Latin America and the southern USA, different species of Triatominae with different behaviors can transmit the disease, and they go by different common names. Here in the USA, they are commonly known as "kissing bugs." They are blood-sucking bugs with mostly nocturnal habits; the parasites are present in the insect's feces and as the bug feeds, it simultaneously defecates on the person (or animal) that is biting. Then, the parasite can infect the host through the lesion left behind by the bite. It doesn't seem very efficient, and yet it infects 5 million people in Latin America, according to the last regional epidemiological update by WHO, which is based on 10-year-old data. There are other ways in which people can get infected with the parasite: from mother to child during pregnancy, blood transfusion and through food contaminated with triatomine feces. In endemic areas, triatomine insects remain the main transmission route. Chagas disease presents a disproportionately high disease burden on indigenous communities and poor rural peasants in the Gran Chaco eco-region extending over Argentina, Bolivia and Paraguay, where approximately 60 percent of infected people from Latin America come from. Particularly, a third of new cases transmitted by the triatomine vector in endemic areas occur in Bolivia and Argentina. During 2012-2017, I focused my dissertation work in this region, more specifically in rural communities of the Argentine Chaco region, mostly inhabited by indigenous communities. Although in the Argentine Chaco the overall prevalence of human infection with the parasite that causes Chagas disease has declined over the last 60 years, it remains high (27.8-71.1 percent) in rural communities. Access to diagnosis and treatment is one of the remaining challenges for sustainable control of Chagas disease in endemic areas, but also sustainable control of the vector needs to be achieved to prevent future infections. We knew that there was a strong and positive association between vector abundance in the houses and the risk of infection in the householders, but how was this modified by their socio-economic conditions? And how can we use this information to better direct vector control actions and active case detection? Those are some of the questions that I aimed to answer in this research project. The first challenge was to measure socio-economic status in communities under structural poverty. Understanding poverty as a dynamic and multidimensional process (as opposed to a merely lack of resources) requires introducing the concept of social vulnerability, which considers the "defenselessness, insecurity, and exposure to risks, shocks and stress" experienced by households. However, in the context of low- and middle-income countries, socio-economic inequalities have been studied using proxy indicators such as educational attainment and household ownership of assets, which at best partially capture the full complexity of poverty. As a first step, we developed a social vulnerability index using methods from economics to measure the socio-economic status of the household. In an article published in Parasites and Vectors, we found that indigenous and migrant households had a higher social vulnerability compared to non-indigenous households, and that was associated with an increase in infected vectors in the house. As a second step, we wanted to know how the vector abundance and the social vulnerability interacted to determine the actual risk of infection for householders. For this purpose, we conducted blood tests in the population, and we found that infection was more prevalent in indigenous people compared to non-indigenous people and that it increased both with the abundance of infected vectors and with household social vulnerability. We also found that the social factors modulated the effect of the abundance of infected vectors; vulnerable-household residents were exposed to a higher risk of infection even at low infected-vector abundance, and human mobility within the area determined a lower and more variable exposure to the vector over time. These results are described in a recent paper published in PloS NTD. As a final step, we integrated these results in a risk map that showed high-priority areas. The approach we used can help identify the groups that are most at risk within apparently uniformly impoverished rural communities. The social vulnerability index may be adapted to identify the most vulnerable households affected by multiple health burdens.  Map showing areas at high risk for Chagas disease in the Argentine Chaco region. Image courtesy Maria del Pilar Fernandez Although the approach proposed here can be applied more broadly, there is no universal protocol of intervention with respect to the determinants of health of NTDs. Intervention strategies oriented to reduce the impact of NTDs must be tailored to specific social contexts, capacities and resources available, in order to maximize their impact and cost-effectiveness. The synthetic approach that we used to assess socio-economic inequalities provides key information to tailor and guide targeted vector control actions, case detection and treatment of Chagas disease, and facilitate the integration with other health burdens, towards sustainability of interventions and greater reduction of health inequalities. My experience working on Chagas disease has taught me that sustainable control of vector-borne transmitted diseases and other environmental health-related issues can only be achieved by thoroughly assessing the multiple biological, ecological, socio-economic, cultural and political factors involved. These are not isolated and independently acting factors but interact within a network involving multiple stakeholders with different perceptions and interests, in ever-changing contexts. As we face old and new challenges in our efforts to reduce the burden of NTDs and decrease health inequalities, globally and at national levels, we need to reflect on the lessons learned and the long-standing legacy of the pioneers in the field. As an early career researcher, I want to remain optimistic, and I hope that these efforts of putting NTDs in the spotlight will help us advance towards the Sustainable Development Goals by 2030. Maria del Pilar Fernandez is a disease ecologist specializing on vector-borne disease transmission and a current Earth Institute postdoctoral fellow. Her research focuses on integrating traditional epidemiological research with an expanded perspective including eco-bio-social determinants, their eventual interactions and spatial patterns, through mathematical models. Her ultimate goal is to identify critical factors affecting disease transmission, which will aid in the design of improved intervention strategies to alleviate the biological and socio-economic burden of these diseases in affected communities. The results of the Chagas disease research presented here stem from a broader long-term research program on the eco-epidemiology and control of Chagas disease in the municipality of Pampa del Indio, a highly endemic, mostly rural area of the Argentine Chaco. This project is led by Ricardo E. Gürtler at the University of Buenos Aires – CONICET. M. Sol Gaspe and Paula Sartor were also involved directly in the research presented here. |
| Pathogens in food – why foodborne parasites are relevant too - New Food Posted: 16 Aug 2019 12:00 AM PDT In the summer of 2018 2,299 laboratory-confirmed cases of cyclosporiasis in the US were reported to the Centers for Disease Control and Prevention, with at least 160 people hospitalised. Professor Lucy Robertson, Chair of a COST Action on foodborne parasites, along with Working Group leaders from the Action, explains why foodborne parasites are important – and why they remain the neglected pathogens, despite their significant impact.  The increased incidence of cyclosporiasis in the US last year was not a one-off event. In 2017 there were 1,065 laboratory-confirmed cases in the US, with 384 being reported in 2016, and 546 reported in 2015. In 2014 there were 304 cases, while 2013 was another big year, with 631. Although the infection sources are often not identified, many cases have been associated with fresh produce, including imported coriander, pre-packaged vegetable trays, mixed salads from a well-known fast-food chain, basil, and various other fresh produce. This particular foodborne parasite (FBP) seems to return to US consumers on an annual basis; but why does this happen and how can we prevent it happening again? Other foodborne parasites making a splash during 2018 In addition to cyclosporiasis bothering US-based consumers in 2018, other foodborne parasitic diseases affected consumer health last year. In Santa Maria, Brazil, an outbreak of toxoplasmosis occurred. Over 800 cases had been registered by October 2018, including 114 in pregnant women and three foetal deaths. Another parasite, Trichinella, caused several disease outbreaks in Argentina that same year; one in Pehuajó resulted in over 160 infections, one in Canals in Córdoba Province resulted in over 230 cases, and in Bahía Blanca over 100 infections were reported in September. Most outbreaks were due to consumption of sausages or salami. Symptoms included fever, severe muscle pain, headache, ocular symptoms, diarrhoea and vomiting. Meanwhile, in spring 2018 the Japanese health ministry issued a warning about the parasite Anisakis in raw fish after several reports of infection in various areas of Japan, with acute epigastric pain being the main symptom. On a smaller scale, in Norway, sharing home-pressed apple juice with work colleagues in the autumn resulted in those colleagues reporting in sick one week later with violent diarrhoea due to infection with Cryptosporidium parvum. This snapshot illustrates the diversity of both symptoms and food vehicles associated with parasites. It also leads us once again to question why parasites are the neglected group among foodborne pathogens, and what their relative importance is. Some of these issues have previously been raised1 and problems specific to FBP that have been highlighted include their complexity, diagnosis and transmission attribution difficulties, and the lack of standard detection methods. We address some of these below, using examples of different parasites that also illustrate the importance and impact of these foodborne pathogens. Foodborne parasites: they're complicatedOne problem with parasites is, perhaps, their enormous diversity. Some are protozoa: minute, single-celled organisms that, like bacteria, can only be seen with a microscope; others (such as Trichinella) are worms, but are also microscopic; while some (like Anisakis) are over 1cm long. Some are even bigger: tapeworms (cestodes) can be several metres in length (see Figure 1), although their transmission stages (the part consumed with food to pass on the infection) are generally smaller. Most important, however, is the fact that FBP lifecycles vary tremendously. Some FBP transfer relatively simply from infected host to infected host by transmission stages excreted in faeces contaminating food. Others, however, may go through one or more intermediate hosts, and the infectious stage may end up in the flesh of a fish or animal, which is later ingested. Thus, to understand how food may contain an infective parasite stage requires knowledge about complex lifecycles. Excellent lifecycle illustrations are available (see, for example, www.cdc.gov/dpdx/az.html) – but it is questionable how often these are consulted by those in the food industry.  Figure 1: Single tapeworm (Taenia saginata) from a patient in Belgium in July 2018 (Photo credit, Idzi Potters, Institute of Tropical Medicine, Antwerp, Belgium. Confusingly, some parasites may have several possible routes of transmission to humans; different food types may be associated with transmission of different life stages of the same parasite, and the resulting symptoms may also differ. This is the case for some protozoan parasites, but is particularly important, although much neglected, for some tapeworms. For example, the pork tapeworm, Taenia solium, may infect humans when undercooked pork from an infected pig is consumed; cysticerci (tapeworm larvae) in the pork may result in a tapeworm developing in the intestine of the person. Although this worm may be several metres long and cause abdominal symptoms, the infection is considered relatively mild. However, Taenia solium eggs excreted in the faeces of an infected person may contaminate fresh produce, and if these are consumed then the cysticerci may develop in the unfortunate end consumer. Cysticerci in the central nervous system can cause serious symptoms and the infection may even be fatal. Indeed, Taenia solium is considered to be the most significant FBP globally, causing around 28,000 deaths.2,3 As illustrated, being vegetarian will not prevent infection by FBP parasites. Another example is Toxoplasma gondii. Calculations of disability adjusted life years (DALYs – a metric including both mortality and morbidity), showed that this foodborne pathogen has a high disease burden in the Netherlands,4 and elsewhere.5,6 As with other FBP, this burden is due to chronic, long-term sequelae. Transmission of Toxoplasma to humans can occur during various parasite stages, via such mediums as undercooked meat, fresh produce, or milk (see Figure 2). 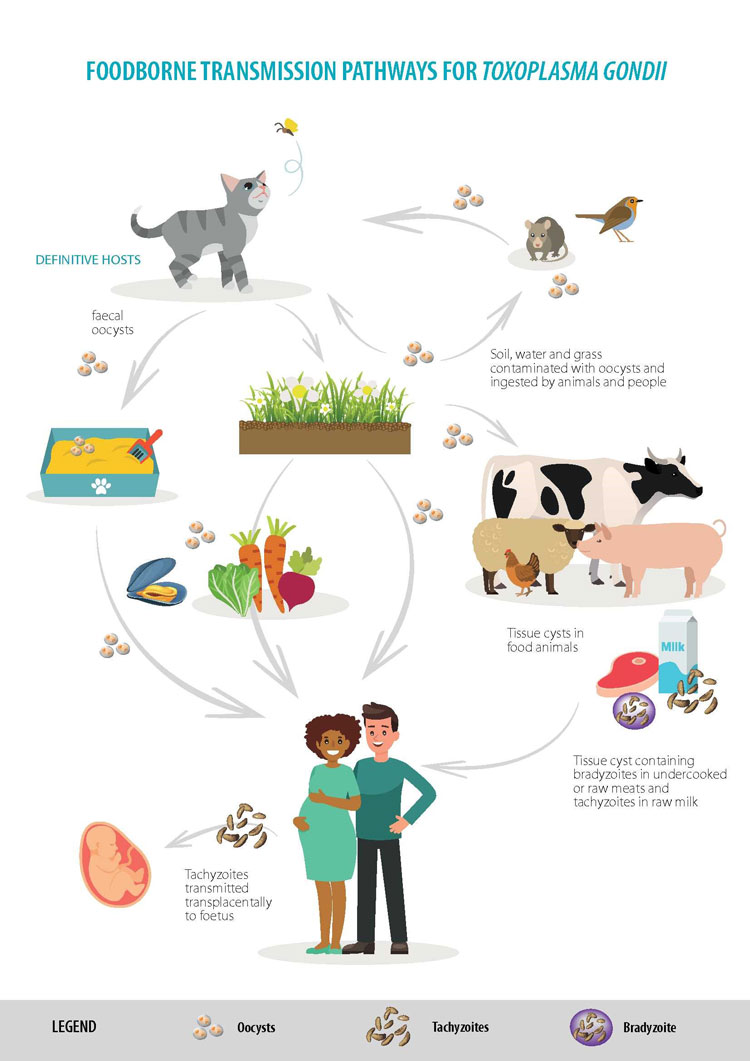 Figure 2: Food transmission pathways for Toxoplasma gondii (Photo credit EFSA, 2018) Foodborne parasites and the slow disease movementOne reason why both consumers and food producers underestimate the likelihood and impact of FBP is due to the often-long period between ingestion of the parasite and onset of symptoms from the resultant infection. This means that pinpointing the meal from where the parasite originated – or, for some parasites, whether it was foodborne at all – is difficult. It is simply hard to make the connection between something that was eaten a while ago and being ill now. Generally, when people blame something they ate for feeling unwell, they point to what they consumed the day before. However, for FBP the period between infection and symptoms can sometimes be several days, but more usually weeks or months, and even years (see Table). Diagnosis may also be delayed, as the symptoms are often diverse and not pathognomonic, or because appropriate diagnostic tests are not requested. For example, opisthorchiasis, which is transmitted to people by ingestion of undercooked freshwater fish, is often asymptomatic, but may cause fever, abdominal pain, diarrhoea, fatigue, and diffuse myalgia. Furthermore, untreated chronic infection may result in the late development, after several years, of cholangiocarcinoma, a malignant cancer of the bile ducts with very poor prognosis. Thus, early diagnosis and appropriate treatment are essential. However, several outbreaks of opisthorchiasis in Italy between 2007 and 2011 indicate that many physicians in Europe are not really aware of this disease. This may result in people living undiagnosed and at risk of developing cholangiocarcinoma.1 In essence, we can expect foodborne parasitic diseases to be underdiagnosed, thus skewing our understanding of the prevalence of these pathogens. Our current awareness of the situation is probably only the tip of the iceberg. Foodborne parasites: we often don't look for them in foodIt may seem logical to look for FBP in food to ensure that it is safe for human consumption, and for some parasites (Trichinella, Taenia saginata cysticerci, Anisakis larvae) in some foods this is indeed the case – although the method sensitivity for some parasites can be low. However, for other FBP, methods to detect parasites in food are either lacking entirely or are impractical and expensive. Legislation that demands that food be checked for FBP is lacking for many FBP. For example, although Toxoplasma is a very important FBP, meat is not examined for this parasite.7 Furthermore, even when outbreaks do occur, implicated produce may not be checked – often due to the prolonged period between infection, symptoms and diagnosis meaning that the implicated food has already been consumed or discarded. This complicates proper source attribution, outbreak investigation, and tracking the origins and routes of infection. For example, an outbreak of cryptosporidiosis in the UK in 2012 involving hundreds of cases was associated with consumption of pre-cut mixed salad leaves, yet suspect foods were not analysed.8 Several reasons were given for this – the main one being that, from the first outbreak, over three weeks were estimated to have elapsed since exposure and, given the short shelf-life of salad, none of the contaminated product was likely to be available for testing. What have we learned and where are we going?Returning to the 2018 outbreaks of cyclosporiasis in USA, one in a recurring tide, it is clear that we still have considerable progress to make regarding FBP and their control. For some foods, salting, drying, freezing, or heating will reduce parasite infectivity, but data are scarce compared to those available for bacteria, and the means to assess parasite viability it is not always obvious.9 For minimally-processed foods or fresh produce, control options are few. This complicates addressing FBP in HACCP-based food safety control systems. With growing trends for eating minimally-processed food products, animals raised outdoors, and organic farming systems, the likelihood of our food containing FBP will likely increase. Fundamental questions remain about the epidemiology, survival and virulence of FBP. In addition, methods for detecting different parasites in various kinds of food, and removal or inactivation of FBP are not optimised. Answering these questions is fundamental, and initiatives to address them are appearing as knowledge gaps are recognised by relevant agencies. However, until both food suppliers and consumers become aware of FBP, finding answers to these questions will be a prolonged process. References
AcknowledgementsThis article was written as a collaboration within the framework of EURO-FBP, A European Network for Foodborne Parasites, COST Action FA1408. About the authorsLucy J. Robertson (Chair of COST Action FA1408) is a Professor in the Department of Food Safety and Infection Biology, Faculty of Veterinary Medicine, Norwegian University of Life Sciences, Adamstuen Campus. Joke WB van der Giessen (Leader of Workgroup 1 of COST Action FA1408) works at the Centre for Infectious Disease Control Netherlands, National Institute for Public Health and the Environment, Bilthoven, the Netherlands. Christian Klotz (Leader of Workgroup 2 of COST Action FA1408) works at the Unit for Mycotic and parasitic agents and mycobacteria, Robert Koch-Institute, Berlin, Germany. Peter Paulsen (Deputy Leader of Workgroup 3 of COST Action FA1408) works at the Institute for Meat Hygiene, University of Veterinary Medicine Vienna, Austria Chiara Trevisan (Deputy Leader of Workgroup 4 of COST Action FA1408) works at the Department of Biomedical Sciences, Institute of Tropical Medicine, Antwerp, Belgium. |
| You are subscribed to email updates from "parasitic diseases examples" - Google News. To stop receiving these emails, you may unsubscribe now. | Email delivery powered by Google |
| Google, 1600 Amphitheatre Parkway, Mountain View, CA 94043, United States | |
Comments
Post a Comment